 
Here we multiplied 1 with 2 because we are calculating the number of electrons in the first shell.So, you can calculate the number of electrons for upcoming shells too. So, for counting the number of lectrons in the first shell we again apply the rule as:. How many electrons should be added in the shell is given by #2n^2# rule where n refers to the number of shell. (b) The energy of the orbit becomes increasingly less negative with increasing n. Draw and explain the atomic structure of Na and K bases on it. 2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. List - IList - III) h v W + K.Ea) Quantization of angular momentumII) E h vb) Wave numbers of Balmer seriesIII) vR2 21 n 21 c) Quantum theoryIV) m.v.r 2h d) Photoelectric effectThe correct match is : Describe the main postulates of the Neils Bohr model of the atom. How do we know how many eletrons should be added to the first shell? In this state the radius of the orbit is also infinite. STRUCTURE FOR SODIUMĮlectronic configuration:- K - 2, L-8, M-1 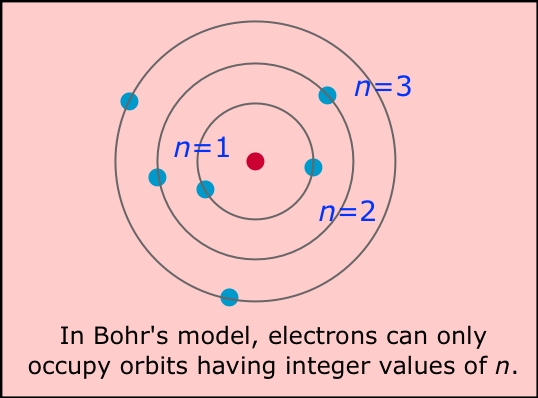
The number of elecrons each shell of an element can old is given by #2n^2# rule given by Bohr where n refers to the number of shell. Diagram representation of the element lithium and sodium illustration. Isomatric of Sodium Atomic Illustration Na 11. Conceptual vector illustration of sodium-23 isotope atom, mass number 23 and electron configuration 2, 8, 1. 
We always do electronic configuration for the number of electrons present in the atom of the element. Bohr model representation of the sodium atom, number 11 and symbol Na. Do the electronic configuration for the atom of the element. Therefore, now you know the number of electrons too. As in every stable atom the number of protons is equal to the number of electrons. Once you know its atomic number you know the number of protons and electrons in it. In an atom, the number of protons the number of electrons the atomic number of that atom. Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist. After him many physicists come forwards with their new ideas and put light on the structure of atom among which Rutherford’s atomic model, Neil Bohr’s and Sommerfeld’s atomic model was taken under consideration. The electron is not allowed to occupy any of the spaces in between the orbits. As per the Bohr model of an atom, the argon atom consists of 18 protons and 22 neutrons in the nucleus while 18 electrons revolve around the nucleus. He was the first who gives us structure of atom. The 3 electrons revolve around the nucleus. According to the Bohr model, or planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. Always write the atomic number of the element first. The Bohr model of the Lithium atom has 3 protons and 4 neutrons in the nucleus. What is the Bohr model for sodium The Structure of an Atom: A Bohr model is a way to show the structure of an atom in regards to its subatomic particles. Atomic number of any element refers to the number of electrons in an atom of that element they are having. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium.Ītomic number of sodium is 11. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero.We draw atomic structures for any element with the help of atomic number they have. \) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
